AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
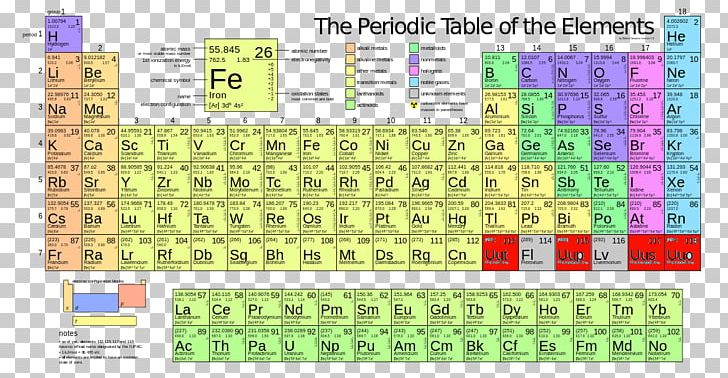
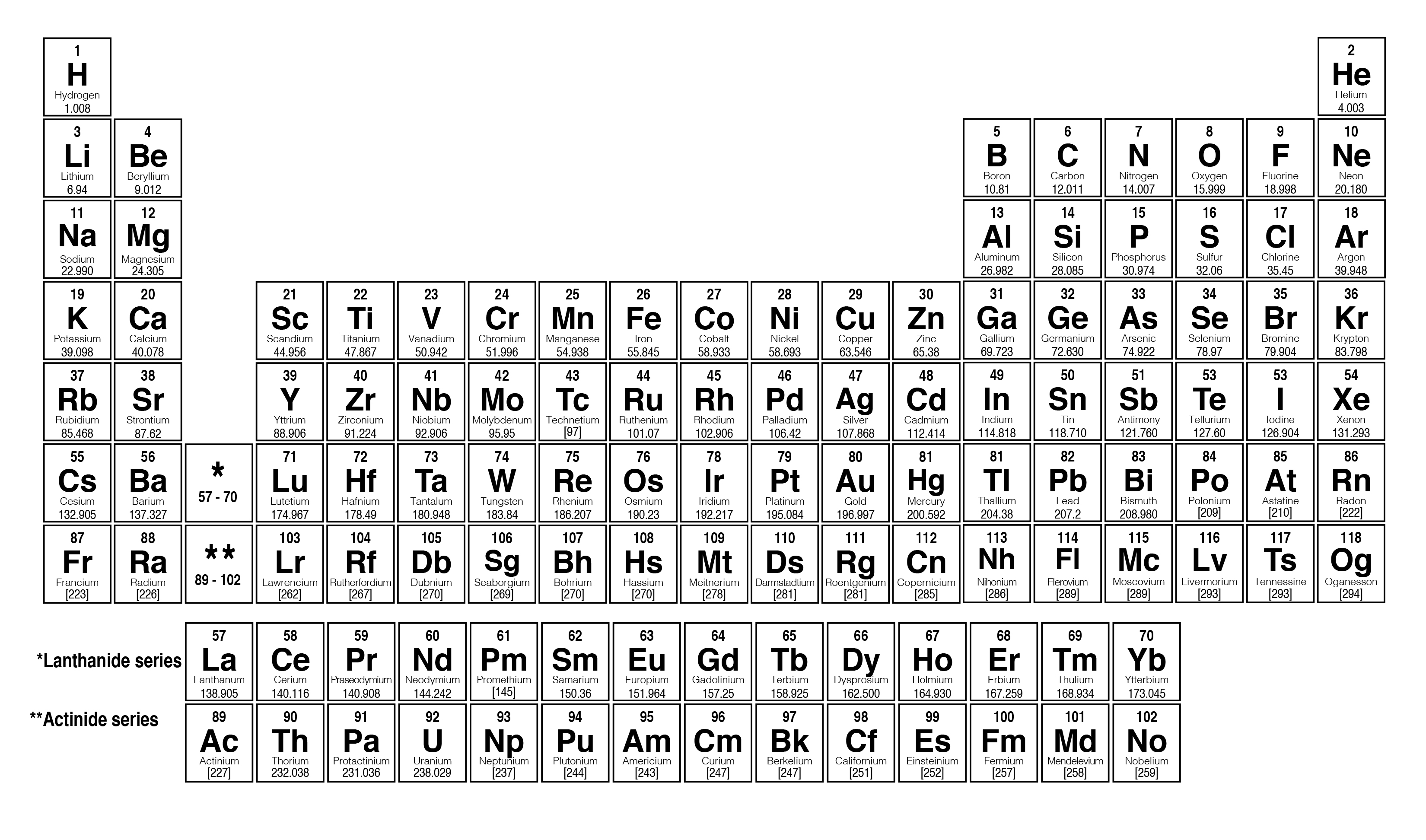
Molar mass from periodic table3/31/2024  
The difference is that molecular mass is the mass of one specific particle or molecule, while the molar mass is an average over many particles or molecules. The molecular mass and formula mass are commonly used as a synonym of molar mass, particularly for molecular compounds however, the most authoritative sources define it differently. The molar mass is appropriate for converting between the mass of a substance and the amount of a substance for bulk quantities. Most commonly, the molar mass is computed from the standard atomic weights and is thus a terrestrial average and a function of the relative abundance of the isotopes of the constituent atoms on Earth. The molar mass is an average of many instances of the compound, which often vary in mass due to the presence of isotopes. The molar mass is a bulk, not molecular, property of a substance.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed